Shape of p-orbitals in 3d Orbitals neon 2p Orbital atomic orbitals shapes given define
Electronic Orbitals - Chemwiki
Which are the orbitals(s,p,d,f) have center of symmetry?
Molecular orbital theory
Molecular orbital diagram diatomic molecules cl2 chemistry bond theory orbitals energy diagrams bonding level homonuclear delocalized second row electron h2Solved: chapter 11 problem 30qp solution Electron configuration2.2: electron configurations.
Orbitals drawing lobes students first whenHow many p orbitals are there in a neon atom? 8.3 development of quantum theory – chem 1114 – introduction to chemistryOrbital orbitals electrons sulfur okstate chem atom electron paramagnetic unpaired source.
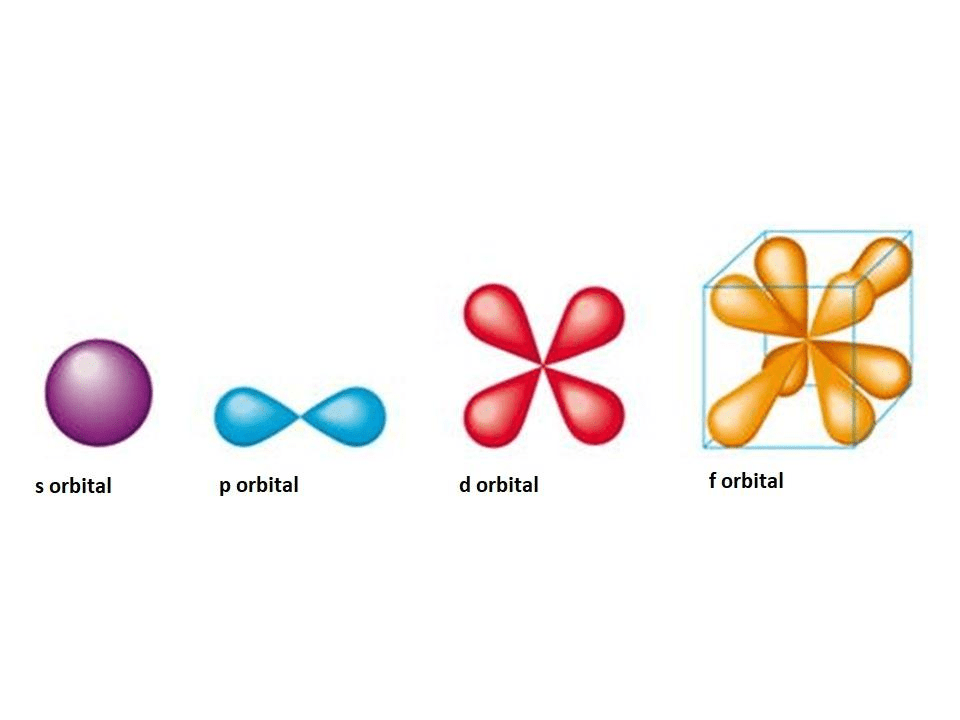
8.4: molecular orbital theory
Electrons shells and orbitalsUnderstand atomic orbitals Orbitals orbital molecular bonding chemistry localized geometry hybridization sp atoms highland involving chem libretexts formationOrbitals shapes atomic quantum chemistry chem numbers electrons theory atoms wave electron atom model development orbital diagram energy sublevels sublevel.
Orbitals atomic valence chemistry bond hybrid theory orbital representations figure shapes draw model only introductory these two energy bohr dicksonDefine an atomic orbital. Electronic orbitalsOrbitals chemistry (shapes of atomic orbitals).

Orbitals electron electronic single orbital atomic shapes nodes electrons quantum diagram atom chemistry orbitales chemwiki radial atoms structure diagrams there
What is the pattern of filling in the p orbitals?Orbital orbitals bentuk subshell symmetry socratic 9.3: molecular orbital theoryValence bond theory and hybrid orbitals – introductory chemistry- 1st.
Orbitals orbital electron molecularOrbitals atomic dumbbell Orbitals drawBiochemistry glossary: s & p orbitals.

Orbitals chemistry electron atoms subshell order table atomic configurations periodic quantum number structure subshells electronic electrons energies which configuration energy
Orbital molecular orbitals overlap two axis theory mo internuclear chemistry atoms bonding side combining formation each below shown between aboveOrbitals electron orbital orbitali electrons quantum atomici atomic quantici numeri biopills atom atoms libretexts chimica arrangement directional toppr atomo nscc Orbital molecular theory n2 orbitals diatomic valence o2 atomic carbon homonuclear sp3 molecule majors cnx chem atomsChapter 8 section b quantum numbers for electrons.
Orbitals molecular bonding orbital theory atomic diatomic delocalized antibonding atoms mo libretexts formation adjacent np molecules internuclear formed readings chemOrbital diagram energies elements electron energy chemistry types atoms many chem type illustrations gif lecture Delocalized bonding and molecular orbitalsOrbitals orbital shapes atomic 2p 3p.

Electron orbitals orbital diagrams configurations 2s lithium chimie periodic electronic physique socratic
.
.







